A) Electronegativity decreases across a row of the periodic table.
B) Electronegativity increases down a column of the periodic table.
C) Electronegativity increases across a row of the periodic table.
D) Electronegativity does not change down a column of the periodic table.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules are constitutional isomers?
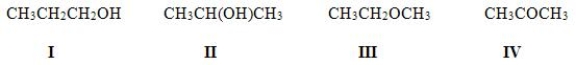
A) I, II, IV
B) II, III, IV
C) I, III, IV
D) I, II, III
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would you expect to have ionic bonds?
A) CO
B) FBr
C) NF3
D) NaCl
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following resonance structures is the least important contributor to the resonance hybrid of the formate anion, HCOO-?
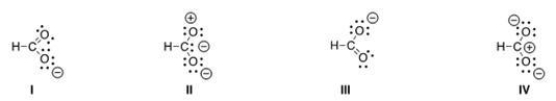
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about resonance structures is true?
A) Resonance structures have the same placement of electrons but different arrangement of atoms.
B) Resonance structures have the same placement of atoms but different arrangement of electrons.
C) Resonance structures have the same placement of atoms and the same arrangement of electrons.
D) Resonance structures have different placement of atoms and different arrangement of electrons.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the ground-state electronic configuration of a carbon atom?
A) 1s2, 2s2, 2p5
B) 1s2, 2s2, 2p2
C) 1s2, 2s2, 2p6
D) 1s2, 2s2, 2p4
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about valence electrons is true?
A) They are the most tightly held electrons.
B) They do not participate in chemical reactions.
C) They are the outermost electrons.
D) They reveal the period number of a second-row element.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the appropriate conversion of (CH3) 4C to a skeletal structure?

A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements correctly describes the typical number of bonds for carbon, nitrogen, and oxygen in most neutral organic molecules?
A) Carbon forms 4 covalent bonds, nitrogen forms 2 covalent bonds, and oxygen forms 3 covalent bonds.
B) Carbon forms 4 covalent bonds, nitrogen forms 3 covalent bonds, and oxygen forms 2 covalent bonds.
C) Carbon forms 4 covalent bonds, nitrogen forms 5 covalent bonds, and oxygen forms 2 covalent bonds.
D) Carbon forms 4 covalent bonds, nitrogen forms 5 covalent bonds, and oxygen forms 4 covalent bonds.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 61 - 69 of 69
Related Exams